Safe and Effective Design of Bioequivalence Study of 400 mg of Imatinib in Normal Healthy Volunteers under Both Fasting and Fed Conditions
PRESENTED TO: BioPharma Services Inc.
PRESENTED BY: J. He, N. Gharavi, H. Somasundaram, U. Patel, R. Chandani, F. Trabelsi.
PURPOSE
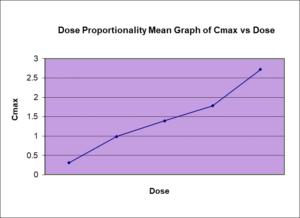
Safe and Effective Design of Bioequivalence Study of 400 mg of Imatinib in Normal Healthy Volunteers under Both Fasting and Fed Conditions – Imatinib is a small molecule protein-tyrosine kinase inhibitor which is used for the treatment of leukemia and stomach tumors. The Food and Drug Agency (FDA) product-specific guidance for bioequivalence (BE) assessment of imatinib mesylate recommends study strength/dose of 400 mg in patients who are already receiving a stable 400 mg dose, rather than normal healthy volunteers (NHV).
In contrast to the FDA, the European Medicines Agency (EMA) has recently issued a product specific BE guidance on imatinib recommending the use of NHV as the study population. EMA considers that a single dose of 400 mg of imatinib could be safely administrated to NHV.
Following extensive safety discussion and investigation, BPSI has conducted many studies with 400 mg imatinib in NHV. The objective of this poster is to share BPSI’s experience from a safety and pharmacokinetic (PK) perspective.
To access the entire publication and view the full resolution PDF, please complete the form.