by The BioPharma Team | May 11, 2022 | Clinical Research - All Blogs, Phase 1, Sponsors
Recruitment of Study Participants Is Vital to Phase 1 & 2 Clinical Trial Success The recruitment and retention of study participants are essential for the success of clinical trials, including Phase 1 clinical trials and 2 studies. Unfortunately, many clinical...
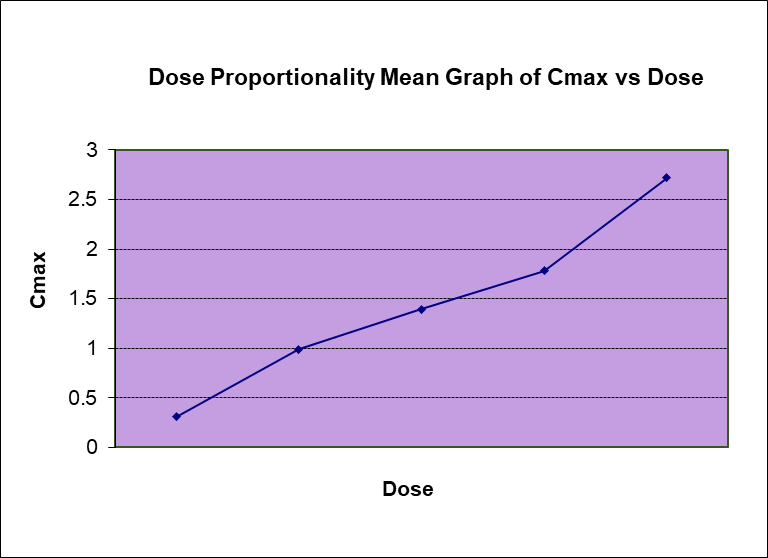
by The BioPharma Team | Apr 28, 2022 | Clinical Research - All Blogs, Phase 1, Sponsors
Dose proportionality: a critical element in drug development and patient therapy In pharmacokinetics, the term dose proportionality is commonly used. If a drug product exhibits dose proportionality, there will be a proportional increase in the amount of drug reaching...

by The BioPharma Team | Mar 31, 2022 | Clinical Research - All Blogs, Phase 1, Sponsors
Our Niche: How Early-Stage Clinical Trial Expertise Delivers Better Client Solutions The process of clinical drug development is complex and associated with high costs and risks. A recent report on drug development found that the overall likelihood for approval of...

by The BioPharma Team | Feb 9, 2022 | Clinical Research - All Blogs, Phase 1, Sponsors
How to Conduct Human Clinical Trials Learn about all aspects of Human Clinical Trials Click to jump to a section: The Importance of Human Clinical Trials Frequently Asked Questions (FAQ’s) About Human Clinical Trials The Phases of Human Clinical Trials...

by The BioPharma Team | Jan 12, 2022 | Clinical Research - All Blogs, Phase 1, Sponsors
Conducting Complex Clinical Studies Involving Speciality Services: an Interview with Louis Co In 2003, when the global outbreak of Severe Acute Respiratory Syndrome (SARS) hit Canada, Louis Co was employed as a nurse in a hospital and a clinical research facility in...

by The BioPharma Team | Jan 6, 2022 | Bioanalysis, Clinical Research - All Blogs, Phase 1, Sponsors
When is 505(b)(2) a Good Choice for Your New Drug Application? Taking a New Drug to Market When you want to market a new drug and there is enough evidence on the drug’s safety and efficacy, the 505(b)(2) is a desirable regulatory pathway for a New Drug Application...








Recent Comments